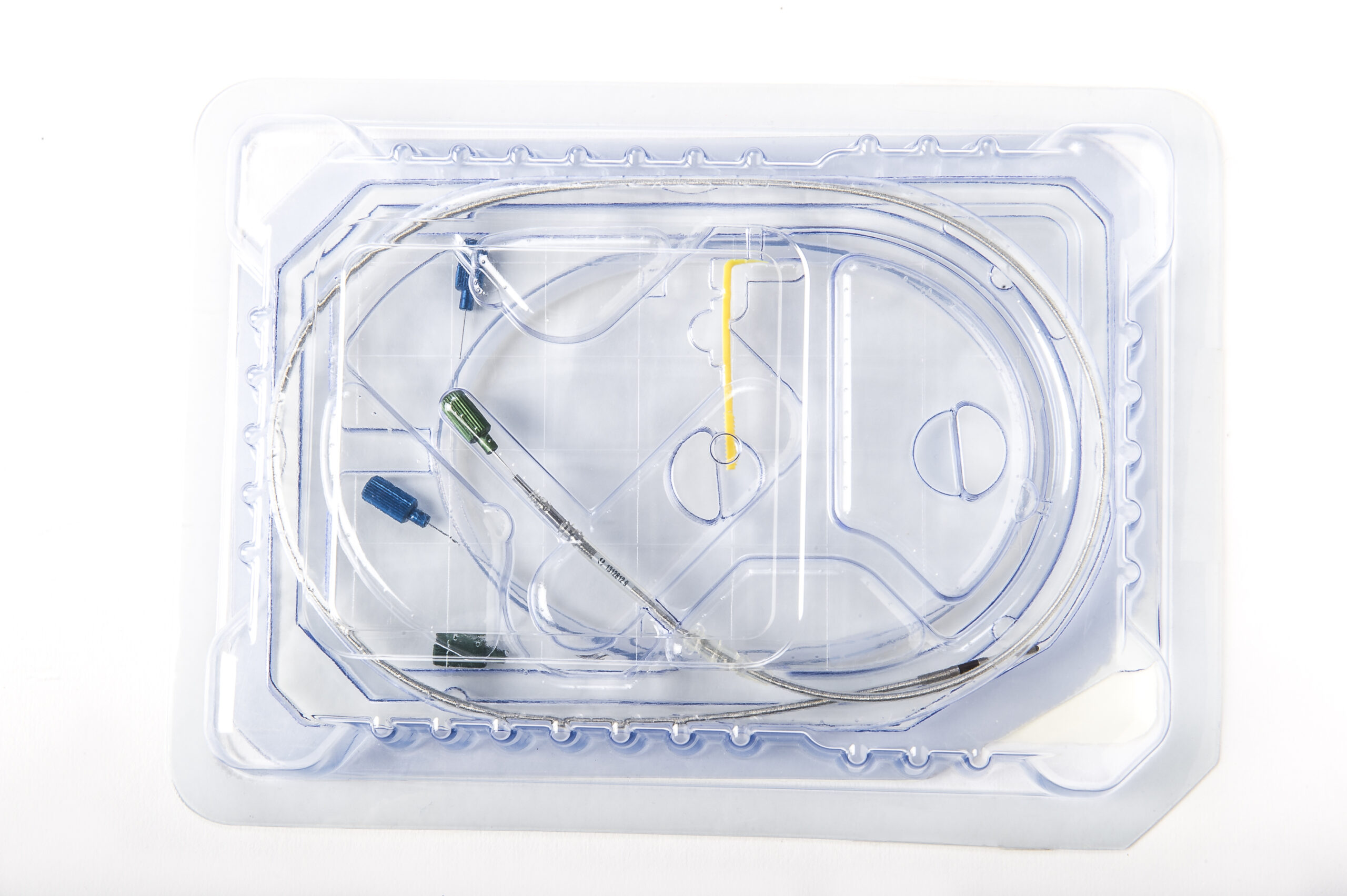
Cardiovascular devices operate within some of the most tightly controlled clinical environments in healthcare. From implantable technologies used during pacemaker surgeries to cardiovascular catheters placed during interventional cardiology procedures, these products must arrive sterile, intact, and immediately usable.
Within those environments, packaging acts as the connection between advanced engineering and clinical use, influencing patient safety, procedural efficiency, and device protection.
As cardiovascular technologies evolve, packaging strategies must adapt to regulatory requirements, validation testing, and complex distribution channels. Understanding how packaging design supports device protection, clinical workflows, and compliance helps cardiovascular device manufacturers reduce risk and maintain consistent performance while protecting patients.
This article explores how effective cardiovascular packaging supports device protection, procedural efficiency, and reliable clinical use—and what to consider when selecting an ISO-certified packaging partner.
Packaging as a Risk-Control Mechanism in Cardiovascular Care
In cardiovascular surgery and interventional cardiology, packaging functions as an embedded risk-control measure. It limits exposure to physical damage, contamination, and handling variability across clinical and logistical environments. This is a high-stakes necessity, as the global cardiovascular devices market is projected to reach $158 billion by 2035, driven by an aging population and a rising demand for minimally invasive procedures.
In that picture, packaging systems are expected to manage risks associated with:
- Product fragility caused by thin-walled polymers, coatings, or flexible geometries
- Vibration, compression, and orientation changes during cardiovascular device shipments
- Sterile barrier compromise during storage and transport
- Human factors during clinical setup and device access.
Injection systems, endoscopic instrumentation, and electrosurgical accessories often require immobilization features to prevent subtle shifts that can affect performance. Even when damage is not visible, micro-stresses can alter device behavior during use. Furthermore, preventing packaging failures is critically important; industry data indicates that packaging issues account for approximately 10% of all medical device recalls, with 31% of those failures specifically linked to holes or breaches in the packaging system.
Additionally, packaging also supports procedural efficiency. Clear labeling systems, optimized tray layouts, and intuitive opening sequences reduce preparation time and minimize handling errors, helping clinical teams work with greater consistency under time pressure.
Material Selection for Cardiovascular Medical Device Packaging
Material selection defines how packaging behaves under sterilization, transport, and long-term storage. Cardiovascular medical devices often rely on combinations of rigid and flexible materials to balance protection with usability. Plastics remain the cornerstone of this sector, estimated to hold a 42.7% share of the medical device packaging market in 2026. Common material considerations include:
- Thermoformed trays for dimensional stability and precise component positioning
- Compression-molded trays for strength in larger or heavier diagnostic equipment
- Multi-layer cartons to support secondary protection and labeling requirements
- High-performance films for sterile barrier packaging compatibility.
Medical device trays frequently use multi-compartment configurations to separate cardiovascular catheters, surgical tools, and accessories. This separation limits contact during transit and supports organized access in clinical environments.
Single-use disposable trays remain common for cardiovascular procedure kits, particularly where infection control and fast turnover matter. At the same time, packaging teams increasingly evaluate mono-material formats to improve compatibility with recycling infrastructure and advanced recycling technologies, addressing long-term concerns tied to healthcare waste streams.
Sterile Packaging and Regulatory Validation
Sterile medical packaging must perform consistently across sterilization, storage, and distribution. Regulatory standards such as ISO 11607 define expectations for sterile barrier packaging systems and validation activities.
Validation programs typically address:
- Seal strength and package integrity testing
- Transit testing simulating vibration, drop, and compression events
- Shelf-life validation supporting labeled expiration dates
- Material compatibility with sterilization methods.
Ethylene oxide and gamma radiation remain widely used sterilization methods for cardiovascular devices, and the packaging materials must tolerate exposure without embrittlement, discoloration, or barrier degradation.

Cleanroom Assembly and Packaging Controls
Once packaging materials and validation requirements are defined, controlled production environments become the next critical factor. Assembly and packaging of cardiovascular medical devices commonly occur within Class 7 cleanrooms built to ISO 14644-1. These environments support sterile workflows while maintaining throughput for high-volume production.
Cleanroom controls usually include:
- Environmental monitoring for particulate levels
- Defined gowning and material entry protocols
- Segregated workflows for sterile and non-sterile operations
- Documented inspection and release procedures.
Effective project management coordinates incoming components, packaging materials, inspections, and documentation. This coordination is especially important when original equipment manufacturers rely on ISO-certified vendors to align production schedules with regulatory requirements and market demand.
Packaging Design Across Cardiovascular Product Categories
Cardiovascular device portfolios rarely follow a single form factor, and packaging design must adjust accordingly.
Examples include:
- ECG patches, where wearability and label clarity matter
- Implantable technologies, requiring rigid protection and extended shelf-life validation
- Diagnostic equipment, often larger and heavier, influencing tray and carton design
- Robot-assisted surgery systems, which introduce complex component sets.
Procedure Kits used in cardiovascular surgery frequently combine surgical preparation kits, electrosurgical accessories, and single-use devices. Medical Device Packaging for these kits must balance accessibility, sterility, and efficient staging within operating rooms.
Packaging strategies must also account for distribution channels. Hospital bulk shipments, distributor fulfillment, and direct-to-consumer shipments expose packaging to different stress profiles, making adaptability an essential design requirement rather than a secondary consideration.
Regulatory Expertise and Compliance Alignment
Regulatory compliance shapes every stage of cardiovascular packaging programs. ISO 13485 quality systems govern documentation, supplier qualification, and change management across assembly and packaging operations.
Packaging programs must align with:
- FDA requirements for labeling systems and traceability
- Regulatory standards governing validation and revalidation
- Controlled change processes for materials or design updates.
Even minor packaging changes can trigger validation activity. With the U.S. market growing at a 7.1% CAGR, strong regulatory expertise is the only way for medical device companies to manage transitions without disrupting supply chain continuity.
Conclusion
As cardiovascular devices continue to move toward greater complexity, tighter tolerances, and different materials, packaging has become an important design and manufacturing decision that directly affects downstream success. Choices made at the packaging level influence how smoothly products move through validation, how reliably they scale into production, and how confidently they are handled in clinical settings.
For medical device manufacturers, the challenge is rarely a single requirement, but rather the accumulation of regulatory expectations, operational constraints, sterilization considerations, and lifecycle changes that must remain aligned over time. Packaging programs that are built with this long view are better positioned to absorb change without disruption.
What helps in the process is working with an ISO-certified partner that understands cardiovascular devices from early development through sustained production. That allows teams to focus on innovation while maintaining control over risk.
When packaging is treated as an integrated part of the manufacturing system rather than a final step, it becomes a stabilizing force—supporting reliable launches, consistent quality, and long-term program continuity.
How PRO-TECH Design Supports Cardiovascular Packaging Programs
As an ISO 13485–certified contract manufacturing organization, PRO-TECH Design supports cardiovascular packaging programs through controlled, repeatable processes designed specifically for regulated medical devices.
Support for cardiovascular programs typically includes:
- Assembly and packaging performed in Class 7 cleanrooms aligned with ISO 14644-1
- Sterile medical packaging validated in accordance with ISO 11607
- Coordination with ethylene oxide and gamma radiation sterilization methods
- Packaging solutions designed to support both high-volume production and specialized device programs.
For medical device manufacturers and original equipment manufacturers, this integrated approach allows packaging programs to remain reliable under regulatory scrutiny while adapting to operational and market-driven change. If you are evaluating packaging solutions for cardiovascular devices or planning a transition to an ISO-certified partner, reach out to PRO-TECH Design. Our team of experienced professionals is there to support the process—from early packaging design through validated, production-ready execution.
FAQs
What makes packaging for cardiovascular devices different from other medical devices?
Cardiovascular devices often exhibit higher product fragility, longer geometries, and tighter tolerances. Packaging must stabilize components while preserving sterile barrier performance.
How does ISO 11607 affect cardiovascular packaging validation?
ISO 11607 defines requirements for sterile packaging systems, including transit testing, shelf-life validation, and material qualification for cardiovascular medical devices.
Which sterilization methods are most common for cardiovascular devices?
Ethylene oxide and gamma radiation are widely used. Packaging materials must maintain integrity and barrier performance after exposure.
Why are Class 7 cleanrooms used for cardiovascular assembly and packaging?
Class 7 cleanrooms built to ISO 14644-1 limit particulate contamination while supporting scalable, high-volume manufacturing.
How does packaging support procedural efficiency?
Organized trays, clear labeling systems, and predictable opening sequences reduce preparation time and handling errors during cardiovascular procedures.
How do distribution channels influence packaging design?
Packaging must perform consistently across hospitals, distributors, and direct-to-consumer shipments, each introducing different handling and transport risks.